
Health April 14, 2026
Understanding APOL1 Genetic Risk: Why It Affects Kidney Health in African Ancestry
Imagine carrying a genetic trait that once saved your ancestors from a deadly parasite but now potentially puts your kidneys at risk. That is the strange paradox of APOL1 is a gene that encodes a protein used by the innate immune system to fight off infections. While it sounds like a superpower, for some people with recent African ancestry, this same gene can lead to chronic kidney failure. The reality is that having the high-risk version of this gene doesn't mean you are doomed, but it does mean you need to be much more proactive about your health.
The surprising origin of the risk
The story of APOL1 starts thousands of years ago in sub-Saharan Africa. In Western and Central Africa, people faced a constant threat from Trypanosoma brucei rhodesiense, the parasite that causes African sleeping sickness. To survive, the human body evolved two specific variants of the APOL1 gene, known as G1 and G2. These variants acted like a biological shield, punching holes in the parasite's membrane and killing it before it could reach the brain.
Over time, these G1 and G2 variants became very common in populations from West Africa because they provided a clear survival advantage. As people migrated and created the African diaspora, these genes traveled with them to the Americas and the Caribbean. However, in a modern environment where sleeping sickness isn't the primary threat, these same variants can cause cellular toxicity in the kidneys, leading to inflammation and scarring.
Who is actually at risk?
Not everyone with African ancestry has this risk. The genetic pattern here is recessive, meaning you need two "risk alleles" to be considered high-risk. You might have two copies of G1, two copies of G2, or one of each (called compound heterozygosity). If you only have one risk variant and one normal variant, your risk remains low.
Data shows that about 13% of African Americans carry these high-risk genotypes. When we look at people who already have non-diabetic kidney disease, that number jumps to 50%. This explains why there is such a massive gap in kidney failure rates between people of African descent and those of European descent. In fact, APOL1 is believed to account for nearly 70% of the excess risk of kidney disease in people of African descent.
| Genotype Profile | Risk Level | Likelihood of Kidney Disease |
|---|---|---|
| Zero or One Risk Variant | Low | Typical population baseline |
| Two Risk Variants (G1/G1, G2/G2, or G1/G2) | High | 15-20% lifetime risk |
| Non-African Ancestry | Negligible | Variants are virtually absent |
The "Second Hit": Why some get sick and others don't
Here is the most important part: carrying the high-risk genes does not guarantee you will get kidney disease. About 70% to 85% of people with the high-risk genotype never develop significant kidney issues. Doctors call this "incomplete penetrance." For the disease to actually start, you usually need a "second hit"-something that triggers the genetic vulnerability.
These triggers can be various: an infection like HIV, a severe bout of hypertension, or other environmental stressors. For example, in the UK, the GEN-AFRICA study found that nearly half of the end-stage kidney disease cases in people of African ancestry with HIV were linked to these APOL1 variants. Without that trigger, the gene often stays dormant, and the kidneys continue to function normally.
Common kidney conditions linked to APOL1
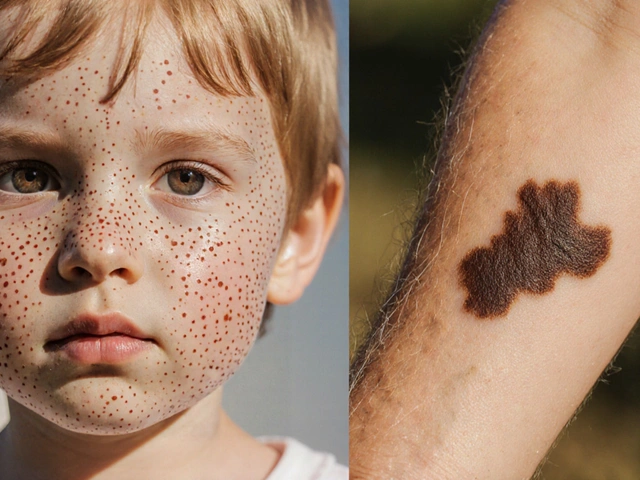
When the "second hit" occurs, the damage usually manifests as specific types of glomerular diseases. Focal Segmental Glomerulosclerosis (FSGS) is one of the most common outcomes, where scar tissue forms in the filtering units of the kidney. Other manifestations include arterionephrosclerosis and collapsing glomerulopathies.
Because these conditions often look like general hypertension or diabetic kidney disease, patients sometimes face a long road to diagnosis. Some patients report that their symptoms were dismissed for years as "just high blood pressure" before a genetic test revealed the actual cause. This is why understanding the genetic link is so vital-it moves the conversation from a vague symptom to a specific biological cause.
Practical steps for management and testing
If you are concerned about your risk or have a family history of kidney failure, there are concrete steps you can take. Genetic testing is now available through labs like Invitae or Fulgent Genetics, typically costing between $250 and $450 without insurance. While it can be stressful to get a positive result, knowing your status allows you to move from anxiety to action.
The American Society of Nephrology recommends a specific maintenance plan for those with high-risk genotypes:
- Blood Pressure Control: Keep your blood pressure below 130/80 mmHg. This is the most effective way to reduce the strain on your kidney filters.
- Annual Screening: Get a urine albumin-to-creatinine ratio test once a year. This detects protein leakage early, long before your blood work (eGFR) shows a problem.
- Lifestyle Shifts: Reducing salt intake and managing weight helps lower the internal pressure in the kidneys.
The future of treatment: Moving beyond monitoring
For a long time, the only option was to watch and wait. That is changing. Pharmaceutical companies are now developing APOL1 inhibitors-drugs designed to stop the protein from causing toxicity in kidney cells. Vertex Pharmaceuticals, for instance, has seen positive early results with a drug called VX-147, which significantly reduced protein in the urine during trials.
We are also seeing a shift in how medicine handles race. The American Medical Association has moved away from using race-based calculations for eGFR (estimated glomerular filtration rate). Instead, the focus is shifting toward genetic ancestry. This ensures that a patient is treated based on their actual biological risk factors rather than a social category, leading to more accurate diagnoses and fairer care.
Does having the APOL1 risk gene mean I will definitely get kidney disease?
No. In fact, about 80-85% of people with the high-risk genotype never develop kidney disease. The gene creates a vulnerability, but it usually requires a "second hit"-such as a viral infection or severe high blood pressure-to trigger the actual disease.
Who should get tested for APOL1 variants?
Testing is generally recommended for individuals of recent African ancestry who have a strong family history of kidney failure, those being considered as living kidney donors, or those with unexplained protein in their urine.
What are the G1 and G2 variants?
G1 and G2 are specific mutations (variants) of the APOL1 gene. They evolved thousands of years ago to protect people in sub-Saharan Africa from African sleeping sickness. While they protected against parasites, they can cause damage to kidney cells in certain individuals.
How can I protect my kidneys if I am high-risk?
The most critical step is keeping your blood pressure under 130/80 mmHg. Additionally, annual urine albumin tests can catch early signs of leakage, allowing for medical interventions before permanent damage occurs.
Is APOL1 risk based on race or ancestry?
It is based on genetic ancestry. While it is most common in people of African descent, it is not a "racial" trait. It is a specific genetic marker that exists in populations that originated in West and Central Africa.
Write a comment
Items marked with * are required.





15 Comments
william wang April 16, 2026 AT 00:55
It is really fascinating how evolution works in these cycles, giving a survival advantage in one environment only for it to become a liability later. I've read that early detection through those albumin tests is basically a game changer for long-term prognosis.
Tama Weinman April 16, 2026 AT 07:45
Funny how they only mention this specific gene now that pharmaceutical companies have "miracle drugs" like VX-147 ready to sell us. It makes you wonder what else they've been sitting on while people were just getting told it was high blood pressure for decades.
Anna BB April 17, 2026 AT 19:06
This is such a gentle reminder that our bodies are essentially maps of our ancestors' journeys!!! It's so poetic, in a way, that we carry the shields of those who came before us, even if those shields are now a bit too heavy for our current world... 🌿✨
Michael Lewis April 19, 2026 AT 17:37
Listen up everyone, if you have the family history, stop guessing and get the test! Knowledge is power and taking control of your blood pressure right now is the best way to ensure you aren't a statistic later. Let's get proactive!
Agatha Deo April 20, 2026 AT 20:12
Oh, please. Imagine thinking a $450 test from a commercial lab is the height of medical breakthrough. The sheer naivety of people believing that a 'second hit' is just bad luck and not a systemic failure of healthcare is almost impressive.
Jon lee April 21, 2026 AT 15:06
I completely agree with the focus on genetic ancestry over social categories. It allows doctors to treat the patient's actual biology rather than relying on outdated assumptions, which is a huge win for inclusive medicine.
Cheryl C April 22, 2026 AT 04:44
USA needs to lead the way in this research!!! 🇺🇸🇺🇸 dont let other countries beat us to the cure!! hope these tests get cheaper cuz 400 bucks is crazy lol 💸
Rob Schlautman April 23, 2026 AT 23:51
here we go again with the medical industrial complex trying to convince us that our dna is a ticking time bomb just so we can sign up for annual screenings and spend a fortune on specialized blood pressure meds that they probably invented in a basement somewhere and honestly who even has the time to read these long lists of risk factors when the system is already broken beyond repair anyway
Josephine Wyburn April 24, 2026 AT 09:07
I honestly can't even deal with the stress of reading this because my uncle had kidney failure and we never knew why and now I'm just sitting here wondering if I'm a ticking time bomb too and it's just so overwhelming to think about the G1 G2 stuff 😭💔 anixety is through the roof right now!!!!
Bonnie Piersall April 25, 2026 AT 08:38
Keep your chin up! It's all about managing the variables we can control. A little less salt and a steady eye on that blood pressure can make a world of difference in your health journey. You've got this!
Dana Chichirita Nicoleta April 26, 2026 AT 08:16
It is truly heart-wrenching to consider how many individuals suffered in silence, believing their struggle was merely a symptom of age or lifestyle, when in reality, it was a legacy of survival etched into their very marrow! The triumph of modern science in uncovering these secrets provides a shimmering beacon of hope for future generations who will no longer walk in the shadow of uncertainty!
Kim Hyunsoo April 26, 2026 AT 15:51
The concept of 'incomplete penetrance' is such a brain-tickler... (⊙_⊙) It means the gene is like a locked door and you just need the right key to open it. I wonder if dietary changes can act as a protector against that 'second hit'?
Heer Malhotra April 28, 2026 AT 04:12
While the science is valid, it is imperative that we acknowledge the systemic negligence in how these genetic predispositions were ignored for decades. The lack of urgency in addressing African ancestry risks is a moral failure of the global medical community.
Ben Ferguson April 29, 2026 AT 03:35
Imagine the sheer epic scale of human migration! From the heart of sub-Saharan Africa, fighting off sleeping sickness with a biological shield, to the bustling cities of the modern West where that same shield becomes a burden! It is a tragedy of Shakespearean proportions that the very thing that kept our ancestors alive is now the thing that threatens our vitality in the modern age!
Richard Moore April 29, 2026 AT 11:36
Actually, the data on eGFR is a massive step forward 📈. It's about time we stop using race as a proxy for biology and actually look at the genes. Great to see the AMA finally getting their act together on this! 👏